
In many settings, ritonavir-boosted lopinavir (LPV/r)-based regimens are only recommended for infants with reported PMTCT exposure on the assumption that NNRTI-associated mutations rarely occur outside this group. Infants with no PMTCTexposure are at higher risk of infection than PMTCT-exposed infants, but the proportion exposed is a function of population coverage of PMTCT. Although PMTCT dramatically reduces the risk of pediatric HIV infection, it does not entirely prevent transmission. However, these data come predominantly from clinical trials and research cohorts and the frequency of prophylaxis-selected drug resistance in routine programs is less well established. Introduction Nonnucleoside reverse transcriptase inhibitors (NNRTIs) are still recommended as part of prevention of motherto-child transmission (PMTCT) regimens, including option B/Bþ, despite the well described selection of resistance mutations among a large proportion of PMTCT-exposed women and their infected infants. ß 2014 Wolters Kluwer Health | Lippincott Williams & WilkinsĪIDS 2014, 28:1673–1678 Keywords: children, drug resistance, nonnucleoside reverse transcriptase inhibitor resistance, prevention of mother-to-child transmission Our results support the use of protease inhibitor-based first-line regimens in HIV-infected infants and young children regardless of PMTCT history. Unauthorized reproduction of this article is prohibited.Ģ014, Vol 28 No 11 resistance. 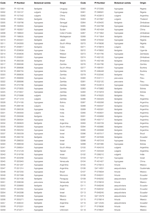
ISSN 0269-9370 Q 2014 Wolters Kluwer Health | Lippincott Williams & WilkinsĬopyright © Lippincott Williams & Wilkins. E-mail: Received: 2 January 2014 revised: 12 February 2014 accepted: 12 February 2014. Sergievsky Center, Columbia University, 630 W 168th Street, New York, NY 10032, USA. Correspondence to Louise Kuhn, PhD, Gertrude H. Sergievsky Center, College of Physicians and Surgeons, and Department of Epidemiology, Mailman School of Public Health, Columbia University, New York, New York, USA, bNational Institute for Communicable Diseases of the National Health Laboratory Services, cFaculty of Health Sciences, Empilweni Services and Research Unit, Department of Paediatrics & Child Health, Rahima Moosa Mother and Child Hospital, University of the Witwatersrand, Johannesburg, South Africa, dMRC Clinical Trial Unit, University College London, London, UK, eHIV Department, World Health Organization, Geneva, Switzerland, fHIV Center for Clinical and Behavioral Studies in the Division of Gender Sexuality and Health, Department of Psychiatry, Columbia University and the New York State Psychiatric Institute, New York, New York, USA, gFaculty of Health Sciences, Wits Reproductive Health and HIV Institute, University of Witwatersrand, Johannesburg, South Africa, and hICAP, Mailman School of Public Health, and Department of Pediatrics, College of Physicians & Surgeons, Columbia University, New York, New York, USA. PMTCT history is an inadequate means of ruling out pretreatment drugĪ Gertrude H. In this South African setting with high PMTCT coverage, almost a quarter of children with no reported or recorded PMTCT also have drug resistance mutations. Conclusion: The new PMTCT strategies dramatically reduce the number of children who acquire infection, but among those who do become infected, NNRTI resistance prevalence is high. The remaining third (32.6%) had no reported or recorded PMTCT exposures, but resistance to NNRTI was detected in 24.0%, NRTI in 10.7%, and protease inhibitor in 1.3%. NNRTI mutations were strongly related to younger age. 
Among PMTCT-exposed, 56.8% had nonnucleoside reverse transcriptase inhibitor (NNRTI), 14.8% nucleoside reverse transcriptase inhibitor (NRTI), and 1.3% protease inhibitor mutations. Results: Two-thirds (67.4%) of HIV-infected children had been exposed to some form of maternal (89%) and/or infant (97%) PMTCT. By this time, more effective PMTCT regimens, including combination antiretroviral therapy for pregnant women, were being implemented. Methods: HIV drug resistance mutations in plasma virus were ascertained using population sequencing among 230 newly diagnosed HIV-infected children under 2 years of age recruited in Johannesburg, South Africa, during 2011. Abramsh Background: In the era of more efficacious prevention of mother-to-child transmission (PMTCT) regimens, documenting the profile of drug resistance in HIV-infected infants and young children is critical to our efforts to improve care and treatment for children. Mellinsf, Vivian Blackg, Lynn Morrisb and Elaine J. Drug resistance among newly diagnosed HIV-infected children in the era of more efficacious antiretroviral prophylaxis Louise Kuhna, Gillian Huntb, Karl-Gu¨nter Technauc, Ashraf Coovadiac, Johanna Ledwabab, Sam Pickerillc, Martina Penazzatod, Silvia Bertagnolioe, Claude A.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
